Melanoma is a type of cancer which usually starts in the skin. It is a cancer that grows from melanocytes – the skin cells which produce the protective pigment called melanin. Since most melanocytes are found in the skin, this is the commonest site for melanoma. Many but not all melanomas grow from moles.
Understanding the structure of the skin helps to show how melanoma develops.
The skin consists of a thin protective outer layer called the epidermis, and a thicker inner layer, the dermis. Each layer is made of individual “building blocks” called cells, and the two layers are separated by a boundary – the basement membrane.
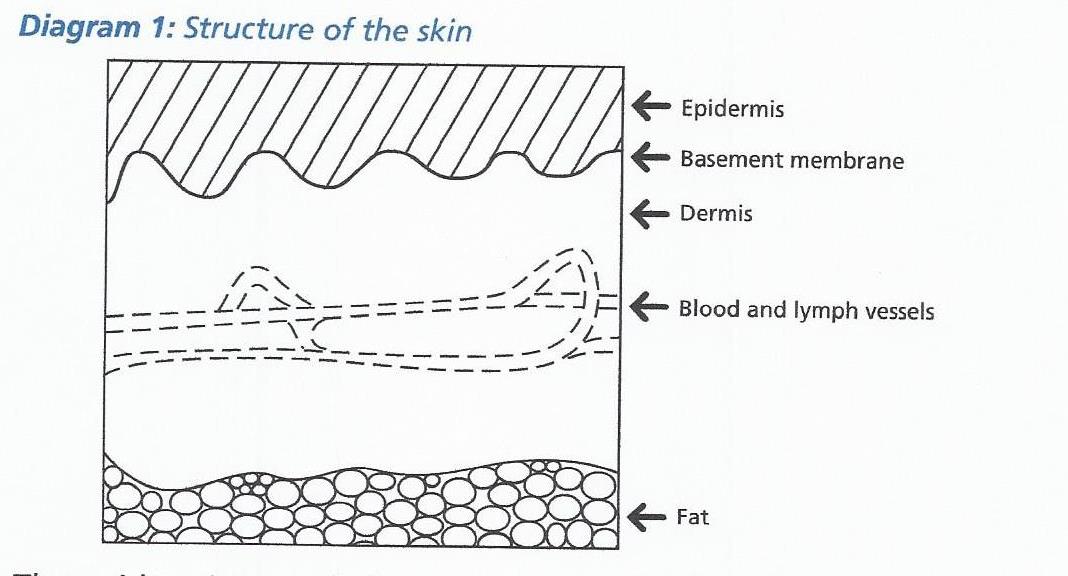
The epidermis is made from two types of cells, called keratinocytes and melanocytes. The keratinocytes make up most of the epidermis, whereas the melanocytes make the pigment that causes the skin to look brown. This pigment is called melanin, and protects the skin from sunburn. The second, inner layer, the dermis, contains blood and lymphatic vessels, which together form the ‘plumbing’ system in the skin.
Melanoma some useful facts:
The skin is constantly wearing out and replacing itself by making new cells. Sometimes things go wrong and the cells replace themselves too quickly. This results in a tumour, which is simply a lump of abnormal cells. Tumours can be benign or malignant.
Benign tumours are usually quite small, they do not grow into the surrounding tissues, and they do not spread elsewhere in the body. For example a wart is a harmless growth of the keratinocytes, and a mole is a harmless growth of the melanoytes in the skin. Benign tumours are not cancer.
Malignant tumours can invade and destroy surrounding tissues and may spread or ‘metastasise’ to other parts of the body. Malignant tumours are cancer.
It is important to remember that cancer is not one disease; it is a group of many diseases. Each type of cancer differs from the others in many ways and the diagnosis, treatment and follow up varies between types of cancer.
Cause of melanoma
The precise cause of melanoma is not fully understood. However there is evidence to suggest that ultraviolet radiation from the sun may damage the skin and cause melanoma. In general the risk is greatest in those with white skin, especially those who burn easily and tan poorly. It is also greater in those with a large number of moles. Melanoma can be inherited but this is very uncommon.
Types of melanoma
Melanomas in the skin occur in two main forms:
- In-situ melanoma
- Invasive melanoma
In-situ melanoma :
This type accounts for about 10% of the melanomas. The most important point about in-situ melanoma is that it is harmless if treated correctly. The cancer cells are confined to the top layer of the skin, the epidermis. They are separated from the blood vessels and lymphatic vessels by the basement membrane, as shown in the picture. However, if untreated, in-situ melanoma can continue to grow and break through the membrane and could then potentially spread. For this reason it is important that they are treated.
Invasive melanoma:
Invasive melanomas are not confined to the top layer of the skin. They grow through the basement membrane into the deeper layer, the dermis. Here, blood and lymph vessels are present which can provide a route for cancer cells to spread around the body. The further down a melanoma has grown into the skin, the greater the chance of the cancer cells getting inside a blood or lymph vessel, and being carried away from the skin to another part of the body. Invasive melanoma is therefore more serious than in-situ melanoma, because the cancer has the potential to spread.
The single most important point about your melanoma is therefore the depth to which it has invaded into the skin, the greater the depth or thickness, the greater the risk of spreading. This measurement is made very carefully on each melanoma that we remove. The measurement is made by an expert pathologist using a microscope. It is measured in millimetres and is referred to as the Breslow thickness (diagram 2).
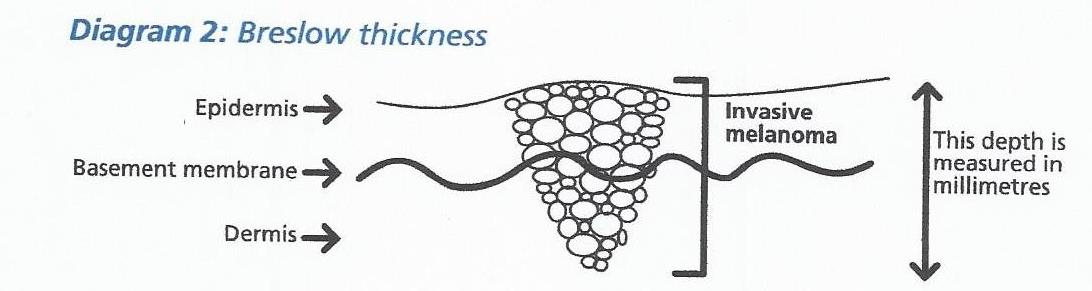
It is important to realise that for most melanomas the chance of spreading is small, because most melanomas remain close to the surface.
The thinner the melanoma, the smaller the chance of it having spread before it was removed, and the greater the chance of cure. If you want to know more about this in detail please ask your Consultant. However, it is important to remember that about 80% of all patients with melanoma are cured.
TREATMENT
Biopsy
Where possible the entire mole or lesion is removed under local anaesthetic, with a margin of normal skin so that it is completely removed. The specimen is sent to the pathology department where it is examined carefully. If it is a melanoma, its thickness, the level of penetration into the skin, and some other features are looked at. This helps us to decide the most appropriate treatment for you.
Melanoma is primarily treated with surgery. Surgical treatments can be divided into three main categories:
- Treatment of the primary melanoma,
- Treatment of the lymph glands
- Treatment of metastases (spread of melanoma). A detailed explanation of the new drug treatments is found further down this page.
PRIMARY MELANOMA
WHY DOES MY SMALL MELANOMA NEED SUCH A BIG EXCISION?
When melanomas grow on the surface of the skin they enlarge in diameter. This happens because the melanoma cells have the ability to move along the surface of the skin. Some melanoma cells move more quickly than others and spread further away from the main tumour. This results in a melanoma which is surrounded by individual scattered cells that are not visible. The deeper a melanoma has penetrated into the skin the wider the scatter on the surface. When treating the primary melanoma it is necessary to ensure that all the invisible scattered cells are removed otherwise the melanoma will re-grow in the same site. This is done by excising (cutting out) a safety margin around the biopsy site of the primary melanoma. This is termed a wide local excision.
The size of safety margin is determined by the depth of the original melanoma because the deeper the melanoma, the wider the scatter effect on the surface. Thin melanomas need a 0.5 cm margin while thicker melanomas require a 2 cm margin of safety. Most of the time the defect that is created with a wide local excision can be closed with a skin flap. In the lower leg, however, there is often not enough stretch in the surrounding skin to be able to do a flap so one has to resort to doing a skin graft.
- LYMPH GLANDS
WHY DO I NEED ALL MY SURROUNDING LYMPH GLANDS REMOVED?
Sentinel lymph node biopsy (SLNB)
When melanomas spread they often do so via the lymph vessels to the lymph glands. For instance a melanoma on the leg would be expected to spread to the lymph glands in the groin. The first lymph gland to be affected is called the sentinel lymph node. It can be identified and removed to test whether the melanoma has spread. It enables us to find out if there has been any spread to the lymph nodes at the time of diagnosis and gives us a more accurate way to stage your disease.
If there is a confirmed metastasis to a lymph node (either an enlarged gland or a positive gland found on sentinel node biopsy) then it is necessary to remove all the remaining lymph nodes from that area (called a therapeutic node dissection or block dissection). This is done because we know that if one gland is affected in an area then there is between a 20% and 40% chance that other glands are also involved. This surgery is performed under general anaesthesia, usually takes about 1-2 hours and patients are required to stay in hospital for one night. If there has been no further spread in the blood, therapeutic node dissection provides an opportunity for cure of the melanoma.
Risks and benefits
As with any surgery there is a risk of bleeding and infection, you may experience some pain and you will be left with a scar. If there is any other risks relating to your surgery the surgeon will discuss this with you before treatment.
After surgery
After your operation you will be given information about any stitches that need to be removed, any dressing changes that are needed and be informed about your follow-up appointment at the rooms. Again this time varies from person to person and may be the following day or could be 2 weeks later.
At this appointment, your wounds will be checked and the pathological results and further management will then be discussed – you may be referred for oncological opinion/management if appropriate.
- SYSTEMIC METASTASES
MY MELANOMA HAS SPREAD. WHAT TREATMENTS ARE AVAILABLE?
If a melanoma has managed to spread in the blood stream to a distant organ (systemic metastasis) surgery remains the best chance of a cure. Even if a cure is not achieved, the survival time with surgery is significantly longer than that obtained with a combination of treatment drugs and radiotherapy. Where possible, surgery is offered as a treatment for spread to organs. Only if surgery is not technically possible or if there are too many sites of spread or one has a high risk of developing metastases, does one consider the need for treatment drugs or radiotherapy.
RADIOTHERAPY:
In terms of radiotherapy, radiation can be used in the adjuvant setting after surgery. When a melanoma is in a clinical site that is difficult to get a wide clearance and adequate margins after surgery, then radiation can be used postoperatively if the wide local excision is narrower than the ideal. Patients sometimes have to undergo removal of lymph glands from certain anatomical sites such as the groin or under the arm and when certain criteria are met, such as many involved nodes or extracapsular spread, patients require postoperative radiation to these areas in order to reduce the risk of local recurrence. This radiation does not improve survival but rather local control. Radiation also plays an important role in palliation or relief of symptoms in patients who have advanced or metastatic disease where the cancer has spread to other sites within the body. It can be used for pain relief or to reduce pressure on structures or to reduce bleeding. Radiation is generally given on a daily basis on weekdays and takes about 15 to 20 minutes to administer and the treatment is aimed and targeted at the area of interest.
NEW DRUG TREATMENTS FOR MELANOMA
All the new treatments for metastatic melanoma are very expensive and most are not covered by medical aids. Only one of the six drugs discussed below has been released for use in South Africa ,namely Ipilimumab. The average cost of each drug is about R1 million per year. The optimal length of treatment for each of the drugs has not been established so no one knows how long they need to be taken for. Some of these drugs have a lot of side effects which forces people to stop taking them and the number of side effects often increases when one combines drugs. Otherwise, the new drugs for treating metastatic melanoma have been able to produce some spectacular results and have brought a lot of hope to many people. There is evidence that some patients may actually be cured of metastatic melanoma by these new drugs.
Research is ongoing and is actually accelerating in the field of biological drugs, so one would predict that many new, more effective and possibly cheaper drugs may be available in the coming years.
SUMMARY OF THE NEW DRUG TREATMENTS FOR MELANOMA
There are two main groups of drugs (all discussed in detail below)
- Chemical drugs that block mutated enzymes in melanoma cells. The mutated enzymes send signals for the melanoma cells to divide and spread. When the enzymes are switched off, the melanoma cells die.
- Vemurafenib
- Dabrafenib
- Trametinib
- Combination therapy with Dabrafenib and Trametinib
- Antibodies that stimulate the body’s own immune system to kill melanoma cells.
- CTLA-4 inhibitors – Ipilimumab
- PD1 inhibitors – Nivolumab
- Pembrolizumab
- Combination therapy with both Nivolumab and Ipilimumab (approved by the FDA in October 2015)
HOW THE IMMUNE SYSTEM WORKS
The main cells of the immune system that are responsible for killing cancer cells are called T-lymphocytes (They get their name from the fact that they are mostly found in lymph tissue. They are called T lymphocytes because they mature in the Thymus gland in the neck). T-lymphocytes cannot immediately recognise cancer cells and therefore have to first be “taught” to do so by other cells. The cells that do the teaching are called accessory or antigen presenting cells. These are very large (but not very numerous) cells that have an innate ability to recognise tumour cells as being foreign. When an accessory cell encounters a cancer cell it is able to ingest (phagocytose) this cell and then destroy it by enzymatically breaking it up into lots of little particles (called antigens). The accessory cell then takes the tumour particles and pushes them through onto the surface of its cell membrane where they become attached to proteins called MHC (major histocompatibility complex protein). The accessory cell then shows or “presents” the tumour particles (antigen) combined with the MHC protein to the T-Lymphocytes (hence the accessory cell’s other name of antigen presenting cell). In this way it shows the T-lymphocytes parts of the destroyed cancer cell and thus “teaches” the T-lymphocytes to recognise the cancer cells that need to be killed. The T-lymphocyte actually binds to the surface of the accessory cell for this process to happen. Once released from the accessory cell, the T-lymphocyte has become activated to go and hunt for cancer cells, which it kills by ingestion (phagocytosis).
An interesting thing happens when the T-lymphocyte is attached to the accessory cell. As well as activating the T-lymphocyte cell, the accessory cell also initiates a process to begin inactivation of the T lymphocyte. It does this by switching on a protein on the T-lymphocyte surface called CTLA-4 (cytotoxic T-lymphocyte-antigen 4). Initially this CTLA-4 (off switch) is not very active but over time it becomes more so until it is so active that the T-lymphocyte switches off. The body does this as a control mechanism so that there is a way of regulating the active T-lymphocyte cells. Unfortunately, the switch-off mechanism is often premature and occurs before all the cancer cells have been killed. Once this switch-off mechanism had been identified, intense research resulted in the production of a drug which allowed the switching off mechanism to be blocked. An antibody was produced which binds to the CTLA-4 (off switch) molecule prior to T-lymphocyte binding to the accessory cell. This thus prevents the CTLA-4 (off switch) from being activated by the accessory cell. T-lymphocytes thus become active for much greater periods of time and can kill a far larger number of cancer cells. The name given to the antibody blocker is Ipilimumab.
Further research has found that active T-lymphocytes have yet another surface molecule that allows them to be switched off. The molecule is called PD1 (programmed death receptor 1). Healthy human cells need to be able to protect themselves from active T-lymphocytes so they all have a protein on their cell surfaces called PD-L (programmed cell death ligand). When an active T-lymphocyte comes along and tries to attack a healthy cell, the healthy cell binds its PD-L molecule with the active T-lymphocyte PD1 molecule and the T-lymphocyte gets switched off. The unfortunate thing is that many cancer cells also carry the PD-L molecule. They can therefore also inactivate the T-lymphocytes and protect themselves from destruction. Science has again stepped in to produce an antibody which, when given intravenously, attaches to the PD1 receptor on the surface of the T-lymphocyte. The function of the antibody is to get in the way and make it impossible for the PD-L molecule on cancer cells to attach to the PD1 receptor on the T-lymphocyte. The active T-lymphocyte therefore cannot be switched off. There are two antibodies that have been produced to block PD1 receptors known as Nivolumab and Pembrolizumab.
- Ipilimumab
– This is a CTLA-4 blocker (blocks the off switch)
– Approved by the FDA in March 2011 for the treatment of metastatic melanoma.
– Sold under the trade name of Yervoy
– One of the big problems with Ipilimumab is that only about 10% of patients have a response to the drug.
– Another problem with Ipilimumab is that about 27% of patients experience severe side effects.
– The good news, however, is that some people appear to have been cured by Ipilimumab with a survival rate of 9% at ten years. – So far, no other treatment for metastatic melanoma can boast such good results.
– Ipilimumab is given as an intravenous infusion initially once every three weeks for four times and can be repeated if there is progression of disease more than three months after an initial response.
- Pembrolizumab (Pembro)
– This was the first of the PD1 inhibitors to be approved by the FDA for treating metastatic melanoma. Approval for use given by the FDA in September 2014.
– It is sold under the trade name Keytruda.
– The average number of patients who respond to the drug is about 34% (compared with 10% for Ipilimumab).
– Of the 34% who respond initially, 74% will still be alive at the end of a year.
– There are far fewer and less severe side effects when compared to Ipilimumab.
– Severe side effects occur in 16% of people.
– Given as an intravenous infusion.
- Nivolumab (Nivo)
– This PD1 blocker was approved for use by FDA in December 2014, only a few months after Pembro had been given the green light.
– Trials have shown that the average number of patients to show a response to the drug is about 44% and that 72% of the responders will still be alive at one year.
– Nivolumab also has fewer side effects than Ipilimumab but about the same as Pembrolizumab.
– Overall, Nivolumab is marginally more effective than Pembrolizumab.
– Given as intravenous infusion.
- Combination Nivolumab and Ipilimumab
– It makes sense to combine the use of a PD1 blocker and a CTLA-4 blocker, i.e. use Nivolumab or Pembrolizumab and Ipilimumab at the same time.
– Both T-lymphocyte off switchers are then blocked simultaneously.
– One study has assessed this using Nivolumab and Ipilimumab together.
– The response rate to this combination of drugs was 58% (which is higher than Nivo alone (44%), Pembro alone (34%) and Ipilimumab alone (19%)).
– One year survival data are not yet available.
– The big problem with combination therapy is that the side effects are severe in about 55% of people.
– In September 2015 the FDA gave approval for the use of combination Nivo and Ipilimumab for metastatic melanoma.
ADJUVANT THERAPY
– Adjuvant therapy is defined as giving a treatment for cancer where there is no cancer detectable on screening.
– Adjuvant therapy is given to patients who have a high risk of developing future metastases, either because their original cancer was at an advanced stage or because they have already had metastases which have been surgically removed.
– The aim of adjuvant therapy is to kill any remaining microscopic cancer cells thus preventing them from growing and thereby curing the patient.
– Until recently there was no adjuvant treatment available for melanoma. Nothing had been shown to work.
– In May 2015, however, a paper was published showing that adjuvant Ipilimumab (at a higher dose than that used in the metastatic setting) given to melanoma patients with surgically resected lymph node metastases significantly reduced the rate of subsequent metastases. At three years, 46.5% of those patients who were given Ipilimumab were still clear of melanoma while this was only true of 34.8% of patients who had received the placebo. It is presumed that this will translate into a long term survival for some patients, but that data is currently not available. In October 2015, the FDA approved Ipilimumab as an adjuvant drug for treating melanoma.
